Big Pharma’s R&D productivity crisis
 The business of developing, testing, and marketing new medicines seems to be in a protracted productivity crisis: the number of new medicines invented per billion dollars of money spent on industrial research and development (R&D) has halved every nine years during the last few decades. To improve their return-on-investment, and to fix this apparent ‘innovation gap’, the pharma industry has started to re-evaluate their R&D strategies. They have changed their business models, consolidated their companies via mergers and acquisitions, increased the number of R&D partnerships and outsourced R&D functions, or reorganized their entire R&D system including of information technology infrastructures and back-office facilities.
The business of developing, testing, and marketing new medicines seems to be in a protracted productivity crisis: the number of new medicines invented per billion dollars of money spent on industrial research and development (R&D) has halved every nine years during the last few decades. To improve their return-on-investment, and to fix this apparent ‘innovation gap’, the pharma industry has started to re-evaluate their R&D strategies. They have changed their business models, consolidated their companies via mergers and acquisitions, increased the number of R&D partnerships and outsourced R&D functions, or reorganized their entire R&D system including of information technology infrastructures and back-office facilities.
Other solutions might comprise better understanding of the biology underlying diseases, better validation of drug targets, or the use of biomarkers and companion diagnostics. Here the industry faces major fundamental problems because medicine development processes are full of uncertainty and chance events. Moreover, these processes not only include research activities, but also competence building and organizational learning within pharma companies, which collectively affect R&D efficiency and efficacy. This makes it extremely difficult to determine, let alone measure, R&D productivity in terms of generating new medicines for the marketplace.
Focusing our attention on research activities, and general trends with the pharma industry, we collected empirical information on scientific research outputs of 23 ‘Big Pharma’ companies that define the bulk of the R&D-active ‘innovative pharma’ sector worldwide. The statistical analysis examined the publication trends and patterns during the last 20 years. Our study was conducted in 2017/2018 and funded by Novartis, the pharmaceutical multinational company with headquarters in Basel (Switzerland).
Results of study
Two of the most salient general observations that emerged from our analysis are:
- non-clinical ‘Big Pharma’ research is increasingly carried out in collaboration with partners outside the company;
- focus of pharma research activities differs significantly from global disease burden.
With regards to the first observation, we focused our analysis on early stage R&D activities and asked ourselves the question: has non-clinical ‘discovery’ research in innovative ‘Big Pharma’ companies become more dependent on inputs and collaboration with partners outside the company? Looking at the non-clinical research publications within the Web of Science database that were (co-)produced by Big Pharma researchers, and computing the share of organizational affiliations outside the company, we were able to determine the Big Pharma’s share in the author affiliate addresses of these publications. The publication frequency counts related to the occurrence of unique affiliations in each publication. An affiliate address referring to a subsidiary was counted as a part the ‘ultimate parent’ company. We monitored the annual trend of these publication output counts from 1995 to 2015. Graph 1 shows the main findings.
Graph 1. External collaboration is increasingly common in discovery and pre-clinical research
(% of discovery research and pre-clinical research publications)
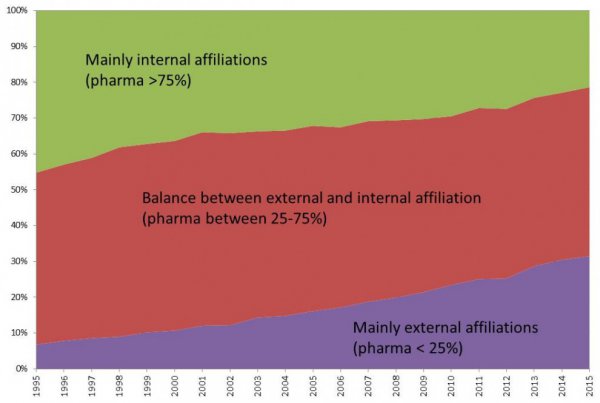
External research cooperation has gradually increased over the years; the share of research publications with a large share of external partners (in the purple-colored zone) has increased fourfold during those two decades - from 7% to 30% of all Big Pharma’s non-clinical research publications. These results are not easily explained, as there are several, interconnected driving forces in play behind the observed trend. On the one hand, how Big Pharma is conducting R&D - where the focus has moved from further developing of in-house R&D capacity to outsource to external research partners, including those in the public sector and with research universities in particular. Trying to benefit from ideas, know-how and resources existing outside the company is one of the strategies to overcome he existing productivity crisis in the Big Pharma sector. On the other hand, during the last years we have witnessed generalized cuts in public R&D investments, which might have led public sector researchers to seek private funding for their academic research. Both these powerful ‘push and pull’ forces created more research cooperation and more co-authored publications. However, the observed upward trend in publication output should be interpreted with due caution because: (a) research cooperation with university partners might be more likely to produce relatively large numbers of publications given academic reward systems and pressures to publish (whereas corporate research may adhere to more restrictive publication practices and remain hidden from the public eye), and (b) academic and/or corporate publication practices have changed during those 20 years.
As for our second observation, the research question was: to what degree is big pharma research activity related to health needs? More specifically: does the distribution of research publications (clinical and nonclinical research) correspond with disease burden as reflected by Disability-Adjusted Life Years? DALYs are a measure of overall disease burden, expressed as the number of years lost due to ill-health, disability or early death. We derived our data from WHO information on worldwide DALYs based on 2012 for selected disease areas, while our worldwide research publication data was based on the years 2009-2013 in the Web of Science. The main results are displayed in Graph 2, which shows major differences between relative research efforts in disease categories (both in pharma and world research) and disease burden in many areas.
Graph 2. Distributions of world’s and Big Pharma’s research publication output does not correspond with disease burden (% share of DALYs and publications across disease areas)
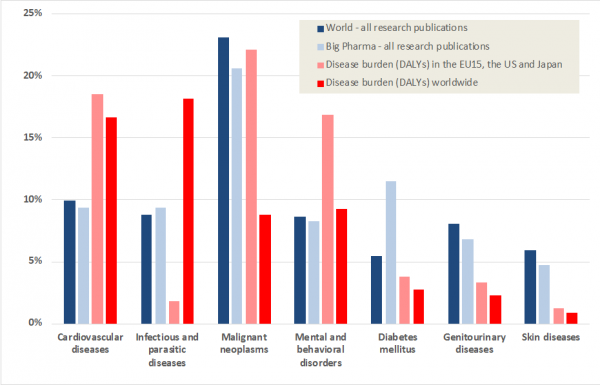
Various factors may explain the apparent under-investment in relation to diseases more prevalent in the low- and medium-income countries of the Global South, such as malaria and tuberculosis, as discussed in a 2013 WHO study based on funding data. First, the fact that most biomedical research is conducted in high-income countries in which these diseases are not considered very important. Second, the fact that the market demand for treatment of these diseases is small, which means that Big Pharma carries out and funds little research on them, indirectly influencing public science. Third, in some areas such as neonatal disease, what appears to be needed is not new medical treatments – which are available, though expensive, in the ‘Global North’ - but research about how to deliver these or other treatments in the healthcare contexts of the lower-income countries of the ‘Global South’. More difficult to explain are the relatively low research efforts regarding stroke or depression in high-income countries (Rafols and Yegros, 2018). Some may argue that this is due to lack of research opportunities, but wider socio-economic influences are likely to play as well an important part (e.g. market demand, stigma in skin diseases, or public visibility in cancer). More detailed analyses of DALYs-related data were recently published as a commentary piece in Nature Index and an online article in La Caixa Foundation.
In summary, the analysis of publication output versus disease burden triggers a variety of questions on current research priorities. We believe that this type of comparisons between health needs and research efforts (which may include complementary sources such as funding data or healthcare costs), can be helpful in health research priority setting, in particular, in initiatives such as the James Lind Alliance’s Priority Setting Partnerships, where a range of stakeholders appraise the health problems that should be addressed and the most useful strategies to tackle them.

